The purity of this Powder Vitamin B raw material is ≥ 99.5, conforms to USP43 and EP10 pharmacopoeia standards, adopts 80 mesh micronization process, and the dissolution speed is increased by 40%. It is specially designed for dietary supplements and functional food processing to meet large-scale demand.
≥ 99.5%
Content
100 over 80 mesh
Number of mesh
≤ 0.5%
Loss on drying


This Powder Vitamin B strictly follows USP43, EP10 and BP2021 pharmacopoeia standard production. Through high performance liquid chromatography detection, the main content is stable between 99.0 and 100.5, and the total amount of heavy metals is controlled below 10ppm, ensuring that the end product successfully passes the FDA and EU compliance review.

For automatic capsule filling and tabletting process optimization, the powder angle of repose is controlled between 30 ° and 35 °. This excellent fluidity allows Powder Vitamin B to be more evenly distributed in the premix, reducing the batch-to-batch mixing coefficient of variation to less than 2%, significantly improving production efficiency.

The particle size of Powder Vitamin B is controlled to D90 < 150 microns using advanced jet milling technology. Compared with ordinary crystalline powder, its specific surface area is increased by 25%, and the dissolution rate in the simulated environment of gastric juice is increased by 40%, which effectively enhances the absorption efficiency of the final product in the human body.

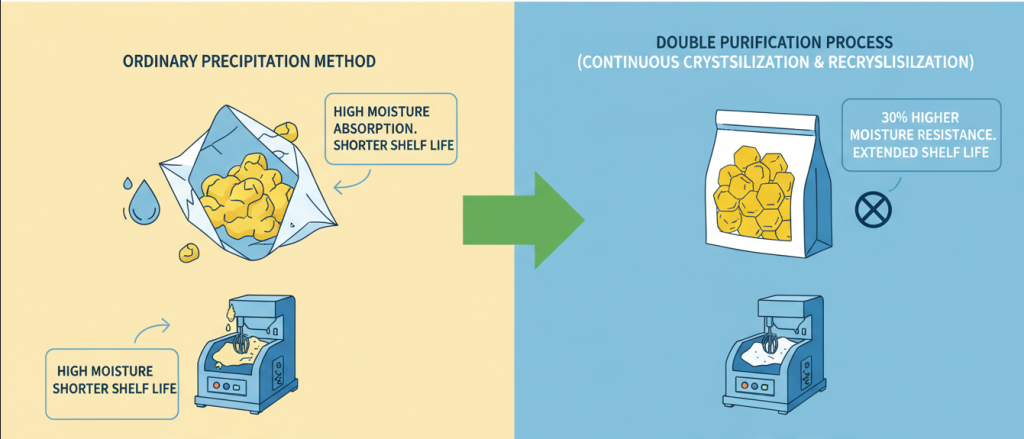
The crystal lattice defects are reduced by the unique crystallization process, so that the Powder Vitamin B is less sensitive to light and oxidation. Under the storage condition of 25 degrees Celsius, the retention rate of the main components is still as high as 98.5 after 24 months, which greatly reduces the risk of loss of distributors in the storage process.

Fluidized bed drying technology is used in the production process to strictly control volatile impurities and moisture within 0.5%. Even in a transportation environment with a relative humidity of 60%, Powder Vitamin B can still maintain a loose powder state, solving the common problem of agglomeration and hardening of raw materials during shipping.

No benzene or chlorinated solvents are used in the extraction and refining process, and the residual solvent content is well below the limit specified in ICH Q3C (e. g. methanol residue <3000ppm). This high-purity Powder Vitamin B is especially suitable for infant formula food and high-end nutrition products with extremely high safety requirements.
| Testing items | Technical Specifications | Detection Method |
| Appearance | White to off-white crystalline powder | Visual inspection |
| Identification reaction | The infrared spectrum is consistent with the reference spectrum. | AND |
| Content determination | 98.5% – 101.0% (on a dry basis) | HPLC |
| pH level | 2.4 – 3.0 | 1% aqueous solution |
| Loss on drying | ≤ 0.5% | Dry at 105℃ to constant weight |
| Burning residue | ≤ 0.1% | USP <281> |
| Total amount of heavy metals | ≤ 10 ppm | USP <231> |
| Lead | ≤ 2 ppm | ICP-MS |
| As | ≤ 1 ppm | ICP-MS |
| Particle size distribution | 100% pass through an 80-mesh sieve | Laser particle size analyzer |
| Bulk density | 0.45 – 0.65 g/ml | Tap density method |
| Microbial limits | Total bacterial count < 1000 CFU/g | USP <61> |



Suitable for factories producing complex vitamin tablets, hard capsules or soft candy. The high-purity Powder Vitamin B ensures a sufficient amount of active ingredient in a limited tablet volume, and its good compression properties are very suitable for high-speed tableting processes.

For businesses that produce energy drinks, fortified flour or nutrition bars. Due to the excellent water solubility and heat resistance of Powder Vitamin B, it can remain stable during thermal processing such as pasteurization and will not cause precipitation or turbidity in the finished product.

For agricultural customers who need to produce high quality premixes. The high fluidity and low electrostatic properties of this product enable it to be evenly mixed with trace mineral carriers to ensure uniform distribution of vitamins in the feed and meet the nutritional needs of modern precision farming.

It is suitable for developing skin care brands with soothing repair effect. Powder Vitamin B’s extremely low heavy metal residue and non-irritating characteristics make it ideal for adding to lotions, creams or shampoos to improve skin barrier function or hair health.


Powder Vitamin B is sensitive to light and heat, and the warehouse should be kept below 25°C and relative humidity below 60%. Be sure to retain the original packaging of the light-shielding layer, after opening the bag if not used up, must be immediately heat-sealed or tied to prevent oxidative degradation.

In the production of solid preparations, it is recommended to use the equal amount of incremental dilution method for mixing. Powder Vitamin B is premixed with the same amount of filler (such as starch or lactose), and then the remaining auxiliary materials are gradually added to ensure the uniform dispersion of trace raw materials in the whole formula.

When preparing liquid preparations, it is recommended to control the solvent water temperature between 30°C and 40°C. Although Powder Vitamin B is readily soluble in water, excessive temperatures (over 60°C) may result in partial loss of potency, and excessive air introduction should be avoided during mixing to reduce oxidation.

Since vitamin B may react with metal ions at a specific pH value, it is recommended that the inner walls of all equipment contacting Powder Vitamin B (such as mixing tanks and storage tanks) should be made of 316L grade stainless steel and be passivated regularly to avoid metal ion catalytic deterioration.

certificate

Welcome to our Frequently Asked Questions page. We have compiled answers to common questions you may have, hoping to provide you with clear and quick solutions. If you cannot find the information you need here, please feel free to contact our customer support team.
The purity of this Powder Vitamin B raw material is ≥ 99.5, conforms to USP43 and EP10 pharmacopoeia standards, adopts 80 mesh micronization process, and the dissolution speed is increased by 40%. It is specially designed for dietary supplements and functional food processing to meet large-scale demand.

Content
≥ 99.5%
Number of mesh
100 over 80 mesh
Loss on drying
≤ 0.5%

This Powder Vitamin B strictly follows USP43, EP10 and BP2021 pharmacopoeia standard production. Through high performance liquid chromatography detection, the main content is stable between 99.0 and 100.5, and the total amount of heavy metals is controlled below 10ppm, ensuring that the end product successfully passes the FDA and EU compliance review.

For automatic capsule filling and tabletting process optimization, the powder angle of repose is controlled between 30 ° and 35 °. This excellent fluidity allows Powder Vitamin B to be more evenly distributed in the premix, reducing the batch-to-batch mixing coefficient of variation to less than 2%, significantly improving production efficiency.

The particle size of Powder Vitamin B is controlled to D90 < 150 microns using advanced jet milling technology. Compared with ordinary crystalline powder, its specific surface area is increased by 25%, and the dissolution rate in the simulated environment of gastric juice is increased by 40%, which effectively enhances the absorption efficiency of the final product in the human body.

The crystal lattice defects are reduced by the unique crystallization process, so that the Powder Vitamin B is less sensitive to light and oxidation. Under the storage condition of 25 degrees Celsius, the retention rate of the main components is still as high as 98.5 after 24 months, which greatly reduces the risk of loss of distributors in the storage process.

Fluidized bed drying technology is used in the production process to strictly control volatile impurities and moisture within 0.5%. Even in a transportation environment with a relative humidity of 60%, Powder Vitamin B can still maintain a loose powder state, solving the common problem of agglomeration and hardening of raw materials during shipping.

No benzene or chlorinated solvents are used in the extraction and refining process, and the residual solvent content is well below the limit specified in ICH Q3C (e. g. methanol residue <3000ppm). This high-purity Powder Vitamin B is especially suitable for infant formula food and high-end nutrition products with extremely high safety requirements.
| Testing items | Technical Specifications | Detection Method |
| Appearance | White to off-white crystalline powder | Visual inspection |
| Identification reaction | The infrared spectrum is consistent with the reference spectrum. | AND |
| Content determination | 98.5% – 101.0% (on a dry basis) | HPLC |
| pH level | 2.4 – 3.0 | 1% aqueous solution |
| Loss on drying | ≤ 0.5% | Dry at 105℃ to constant weight |
| Burning residue | ≤ 0.1% | USP <281> |
| Total amount of heavy metals | ≤ 10 ppm | USP <231> |
| Lead | ≤ 2 ppm | ICP-MS |
| As | ≤ 1 ppm | ICP-MS |
| Particle size distribution | 100% pass through an 80-mesh sieve | Laser particle size analyzer |
| Bulk density | 0.45 – 0.65 g/ml | Tap density method |
| Microbial limits | Total bacterial count < 1000 CFU/g | USP <61> |




Suitable for factories producing complex vitamin tablets, hard capsules or soft candy. The high-purity Powder Vitamin B ensures a sufficient amount of active ingredient in a limited tablet volume, and its good compression properties are very suitable for high-speed tableting processes.

For businesses that produce energy drinks, fortified flour or nutrition bars. Due to the excellent water solubility and heat resistance of Powder Vitamin B, it can remain stable during thermal processing such as pasteurization and will not cause precipitation or turbidity in the finished product.

For agricultural customers who need to produce high quality premixes. The high fluidity and low electrostatic properties of this product enable it to be evenly mixed with trace mineral carriers to ensure uniform distribution of vitamins in the feed and meet the nutritional needs of modern precision farming.

It is suitable for developing skin care brands with soothing repair effect. Powder Vitamin B’s extremely low heavy metal residue and non-irritating characteristics make it ideal for adding to lotions, creams or shampoos to improve skin barrier function or hair health.


Powder Vitamin B is sensitive to light and heat, and the warehouse should be kept below 25°C and relative humidity below 60%. Be sure to retain the original packaging of the light-shielding layer, after opening the bag if not used up, must be immediately heat-sealed or tied to prevent oxidative degradation.

In the production of solid preparations, it is recommended to use the equal amount of incremental dilution method for mixing. Powder Vitamin B is premixed with the same amount of filler (such as starch or lactose), and then the remaining auxiliary materials are gradually added to ensure the uniform dispersion of trace raw materials in the whole formula.

When preparing liquid preparations, it is recommended to control the solvent water temperature between 30°C and 40°C. Although Powder Vitamin B is readily soluble in water, excessive temperatures (over 60°C) may result in partial loss of potency, and excessive air introduction should be avoided during mixing to reduce oxidation.

Since vitamin B may react with metal ions at a specific pH value, it is recommended that the inner walls of all equipment contacting Powder Vitamin B (such as mixing tanks and storage tanks) should be made of 316L grade stainless steel and be passivated regularly to avoid metal ion catalytic deterioration.
