Why Is Glucose Soluble In Water
The core reason glucose dissolves in water is that they are both “polar molecules,” which allows water molecules to form strong attractions called “hydrogen bonds” with glucose molecules. Specifically, a glucose molecule has multiple polar hydroxyl (-OH) groups. The water molecule (H₂O) is also polar, with its oxygen atom being slightly negative and its hydrogen atoms being slightly positive. When glucose enters water, the water molecules, acting like tiny magnets, are attracted to the hydroxyl groups on the glucose molecule and form hydrogen bonds with them. This attraction is powerful enough to overcome the forces holding the glucose molecules together in their solid crystal form. Consequently, individual glucose molecules are “pulled” from the crystal and become evenly dispersed among the water molecules, a process we observe as dissolving.
Polarity Of Molecules
In chemistry, the principle of “like dissolves like” is fundamental and perfectly explains the relationship between glucose and water. Both of them belong to polar molecules, and it is this common ground that determines that they can “accept” each other. The polarity of a molecule, to put it bluntly, is whether its internal charge distribution is uniform. This, in turn, stems from differences in “electronegativity”—the ability of atoms to attract shared electrons in chemical bonds.
Water (H₂O): In a water molecule, oxygen atoms are much more electronegative than the two hydrogen atoms. This means that the bonding electrons will be more biased towards the oxygen atom, giving it a weak negative charge (δ-), while the two hydrogen atoms will be left with a weak positive charge (δ+). This separation of charges, coupled with the fact that the water molecule itself is a V-shaped “curved” structure, makes it a very typical polar molecule with clear positive and negative ends.

Glucose (C₆H₁₂O₆): Although the glucose molecule is much larger, the principle is the same. It has many hydroxyl groups (-OH) in its structure. In each hydroxyl group, oxygen is more electronegative than the hydrogen and carbon attached to it. This results in significant charge separation within each -OH group, making it very polar. With so many highly polar groups on a molecule, the entire glucose molecule naturally becomes a polar molecule.
Hydrogen Bonds
It is precisely because glucose and water are polar that they create the conditions for the formation of a strong intermolecular force—the hydrogen bond. A hydrogen bond is a special, rather strong attraction. It usually occurs between a hydrogen atom that has been bonded to a strongly electronegative atom (such as oxygen, nitrogen, or fluorine) and another nearby electronegative atom.
When solid glucose is placed in water, the water molecules have a weakly positively charged hydrogen end and are strongly attracted to the weakly negatively charged oxygen atoms in the glucose hydroxyl group. Conversely, the weakly negatively charged oxygen end of the water molecule is also attracted to the weakly positively charged hydrogen atoms in the glucose hydroxyl group. This attractive network between these two molecules is the essence of the hydrogen bond in this scenario.
To the forefront of nutrition
Always provide high standards of food additives and nutrition.Delivering innovative solutions that enhance the flavor, texture, and nutritional profile of food,supporting healthier, sustainable products for the food industry.
Dissolution Process
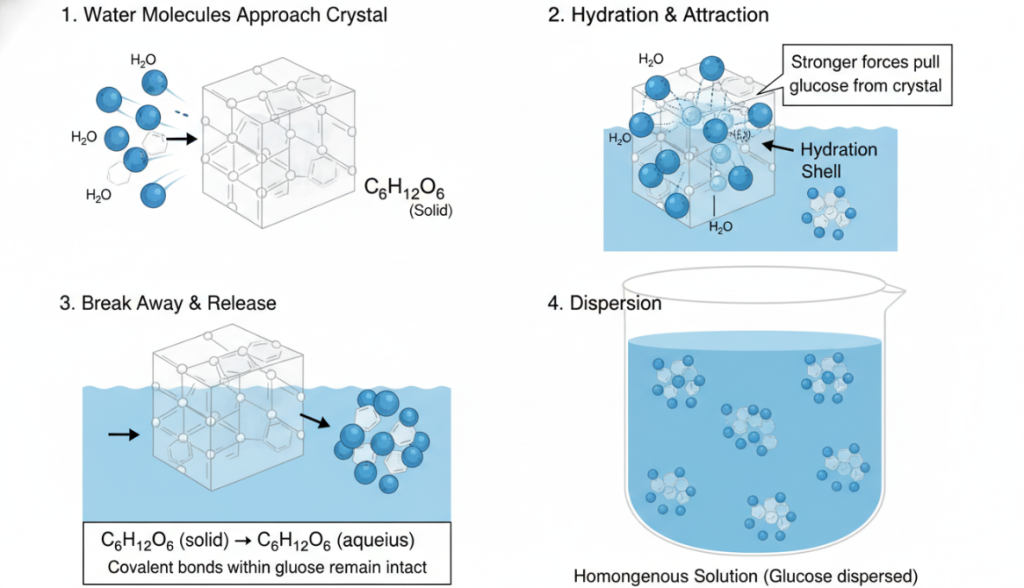
The dissolution of glucose is a dynamic game process at the molecular level.
Water molecules approach the crystals: In the solid state, glucose exists in the form of a crystal lattice, and individual molecules are held to each other by their own intermolecular forces. When this crystal is thrown into the water, the moving water molecules begin to hit its surface.
Hydration and Attraction: Polar water molecules immediately surround the glucose molecules on the surface of the crystal. By forming a large number of hydrogen bonds, the attraction of a group of water molecules to a single glucose molecule becomes stronger than the force between that glucose molecule and its neighbors in the crystal.
Break away and release: This powerful “pull” from water molecules effectively pulls individual glucose molecules out of the solid structure. It must be emphasized here that the water molecule does not break the strong covalent bonds within the glucose molecule; it only breaks the weaker intermolecular forces that maintain the crystal structure. The result is that intact glucose molecules break out of the crystal and go into solution. This process is usually expressed by this equation: C₆H₁₂O₆ (solid) → C₆H₁₂O₆ (aqueous).
Author:Derek
My passion lies in demystifying chemistry. I love taking a simple question, like ‘why does sugar dissolve in water?’, and revealing the elegant molecular science behind it, focusing on core ideas like polarity and the power of the hydrogen bond.
 SGNUTRI
SGNUTRI