What Is Polydextrose Made Of
Polydextrose is actually a highly complex, multi-branched synthetic polymer of glucose. It is produced by random polymerization of three raw materials: approximately 90% corn-derived glucose, 10% sorbitol, and 1% citric acid.
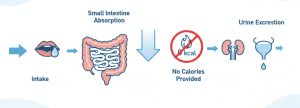
This synthesis process takes place in a high vacuum and high temperature environment, in which citric acid acts as an acid catalyst, promoting the dehydration condensation of glucose and sorbitol, and finally forming a carbohydrate structure with very rich branches. Completely different from those simple sugars, the resulting product contains a large number of 1,6-glycosidic bonds, which are difficult for digestive enzymes in the human body to take apart. This unique chemical composition is the root of what defines it as “water-soluble dietary fiber” and “low-calorie extender”-it provides only one kcal of calories per gram, while showing excellent stability in processed foods.

Three Raw Materials
To fully understand the nature of polydextrose, you have to crack down on the proportions of these three ingredients and the roles they play. Glucose is the backbone of the entire polymer, providing the glucose units needed to build the structure. If there is only glucose, the best that can be tossed out is easily digestible starch or some kind of syrup.
In order to convert it into a non-digestible fiber, sorbitol is added to the formula at a concentration of 10%, which acts as a plasticizer and “caps” the ends of the polymer chains, ensuring that the final product is sufficiently stable and does not crystallize in the finished product. The last 1% of citric acid is the key “engine” of the reaction; as an acid catalyst, it starts the polymerization process without being consumed in large quantities, allowing the glucose molecules to bond in a random, out-of-order manner.
Synthesis Process
The manufacturing process itself is as critical as the raw materials. These raw materials are subjected to extremely severe condensation polymerization under high temperature vacuum conditions.
In this controlled environment, citric acid triggers glucose and sorbitol to lose moisture and fuse together. The molecular structure of this thing is not a linear long chain like flour starch, but a multi-branched spherical structure. Because these chemical bonds are randomly generated, the result is one of connections that the human digestive system does not recognize, which is why the body does not use it as a regular energy source.
Fibres solubles
Fibre alimentaire soluble en gros, adaptée aux compléments alimentaires, aux produits alimentaires, etc.
Prébiotique
Proposer des compléments de fibres alimentaires prébiotiques et des produits de substitution du sucre.
Amidon Sucre
Fabriqué à partir de maltodextrine et de glucose, il convient aux aliments et aux boissons.
Fibre de soja
Les fibres de soja de haute qualité fournissent des matières premières aux industries telles que les compléments alimentaires.
Protéines
Recherche de parasites ultra-faibles et d'intégrité du signal pour les applications à plus de 100 GHz.
Vitamines
Un fabricant de vitamines de haute qualité au service de l'industrie alimentaire et des compléments nutritionnels.
The “Indigestible” Chemical Core
The reason why polydextrose is classified as E1200 rather than sugar is all in its internal structure. Most natural carbohydrates are linked by 1,4-bonds, and tools such as our body’s salivary amylase can cut them off in minutes.
However, the synthesis of polydextrose produces a large number of 1,6-glycosidic linkages. These complex branches are like a “biological shield”. Upon actual ingestion, these bonds are tenaciously resistant to hydrolysis by enzymes in the small intestine. This is the scientific foundation of it as a water-soluble dietary fiber: it can basically pass through the upper digestive tract completely, reach the large intestine smoothly, and play a prebiotic role there.
From Composition To Function
This particular combination of ingredients ultimately results in the one of an extremely powerful ingredient. Because the human body cannot completely metabolize those glucose chains linked by 1,6-bonds, its caloric density drops from 4 kcal/g of ordinary sugar to 1 kcal/g.
For friends who do research and development, this makes polydextrose the first choice to replace sugar and fat. When you remove sugar or fat from the formula in order to reduce sugar, the “weight” and “taste” of the food will instantly collapse; and this specific proportion of polydextrose can fill these physical vacancies without bringing the sugar back.

Résumé
When you ask what polydextrose is made of, the answer lies in the precise, acid-catalyzed fusion of glucose, sorbitol and citric acid. This is actually a microcosm of food science-rearranging simple corn-derived ingredients into a complex, heat-resistant, and low-calorie fibers to accurately respond to the stringent sugar reduction needs of the modern food industry.
Author: Julian Miller
Throughout my career in both the laboratory and clinical settings, I have specialized in the metabolic impact and functional properties of carbohydrate polymers. My mission is to bridge the gap between complex chemical synthesis and nutritional transparency, helping industry professionals and health-conscious consumers understand the science behind every ingredient on the label.
Recommandations connexes
-
Quels sont les effets de l'allulose sur l'organisme ?
194Découvrez comment fonctionne l'allulose, pourquoi il ne fait pas grimper le taux de sucre dans le sang et quels sont ses avantages pour le régime céto, la perte de poids et la santé intestinale.
Voir les détails -
En quoi le processus de production du dextrose et de la maltodextrine diffère-t-il ?
365Comparer l'hydrolyse restreinte et l'hydrolyse complète. Un ingénieur explique les différences entre les processus de production du dextrose et de la maltodextrine.
Voir les détails -
La maltodextrine peut-elle être biologique ?
180Découvrez les avantages de la maltodextrine biologique, les sources sans OGM et les normes d'un véritable régime alimentaire "clean label".
Voir les détails -
De quoi est composé le sirop de glucose ?
385De quoi est composé le sirop de glucose ? Découvrez ses origines végétales, le processus de raffinage et les informations relatives à la sécurité des produits sans gluten.
Voir les détails
 SGNUTRI
SGNUTRI